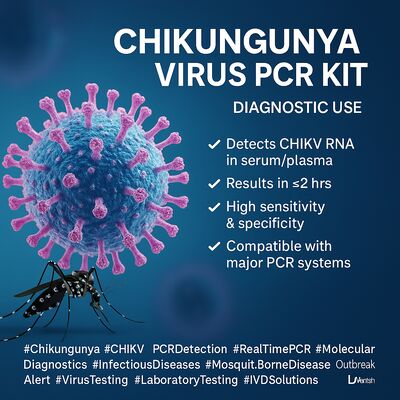
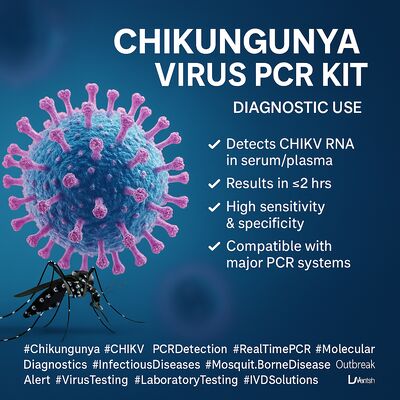
Deja un mensaje
¡Te llamaremos pronto!
 ¡Su mensaje debe tener entre 20 y 3.000 caracteres!
¡Su mensaje debe tener entre 20 y 3.000 caracteres!
 ¡Por favor revise su correo electrónico!
¡Por favor revise su correo electrónico!
PRESENTACIóN
Más información facilita una mejor comunicación.
Sr.
- Sr.
- Sra.
Okay
¡Enviado satisfactoriamente!
¡Te llamaremos pronto!
Okay
Deja un mensaje
¡Te llamaremos pronto!
 ¡Su mensaje debe tener entre 20 y 3.000 caracteres!
¡Su mensaje debe tener entre 20 y 3.000 caracteres!
 ¡Por favor revise su correo electrónico!
¡Por favor revise su correo electrónico!
PRESENTACIóN